Have you ever wondered what happens when you put a jar over a candle? It’s a simple experiment that can reveal surprising things about fire, air, and how they work together.
You might think the candle will just keep burning, but something interesting happens instead. Understanding this can change the way you see everyday things around you. Keep reading, and you’ll discover a fascinating science trick that’s easy to try at home—and it might just blow your mind.
How Candle Flames Work
Candle flames look simple but work through a careful process. A candle burns because of heat, fuel, and air. Understanding how the flame works helps explain what happens when you cover it with a jar.
Each part of the flame has a role. The wax melts first, then turns into gas. This gas mixes with air and burns. The flame stays alive as long as it has oxygen and fuel.
The Role Of Oxygen
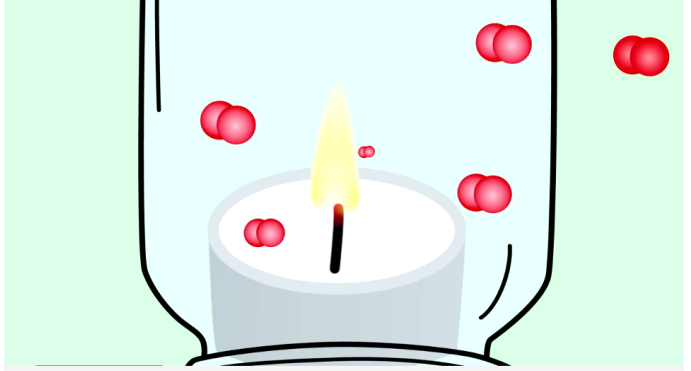
Oxygen is a key part of a candle flame. It helps the wax gas burn and produce light. Without oxygen, the flame cannot keep burning. When you put a jar over a candle, oxygen inside the jar starts to run out.
As oxygen gets low, the flame grows weak. The candle uses the air in the jar quickly. Once oxygen disappears, the flame goes out. This shows how important fresh air is for the flame.
Chemical Reaction In Flames
The candle flame is a chemical reaction called combustion. Wax vapor reacts with oxygen and creates heat and light. This reaction also makes water and carbon dioxide gases.
Heat keeps the wax melting and turning to vapor. The light you see comes from tiny glowing particles in the flame. Without the right mix of wax vapor and oxygen, the reaction stops.
Effects Of Placing A Jar Over A Candle
Placing a jar over a candle creates a small, closed space around the flame. This simple action changes how the candle burns. The air inside the jar is limited and affects the flame’s behavior. Understanding these effects helps us learn more about fire and air.
Watching the candle under a jar shows how fire needs air to keep burning. The changes you see are due to the air inside the jar changing as the candle uses it up.
Oxygen Depletion Inside The Jar
The candle flame uses oxygen to burn. The jar traps the air, so the oxygen inside begins to run out. As the oxygen level drops, the flame gets weaker. Without enough oxygen, the flame cannot stay alive. Eventually, the flame goes out because the air inside no longer supports burning.
This shows how important oxygen is for fire. The jar limits oxygen, making the candle burn only for a short time.
Changes In Flame Behavior
The flame changes shape and size inside the jar. At first, it may burn brighter and taller. This happens because the air is still fresh. Then, the flame starts to shrink and flicker as oxygen runs low. The color of the flame may also change, becoming more yellow or orange. These changes show the flame struggling to get enough oxygen. Finally, the flame dies out completely when the oxygen is gone.
Why The Flame Goes Out
Placing a jar over a candle creates a small, closed space around the flame. This simple action causes the flame to go out quickly. The reason lies in the air inside the jar. The flame needs air to keep burning. Without fresh air, the flame cannot survive.
Lack Of Oxygen
The candle flame needs oxygen to burn. Oxygen reacts with the wax vapor, producing heat and light. When the jar covers the candle, it traps the air inside. The oxygen in the jar is limited. The flame uses up this oxygen fast. Once oxygen runs out, the flame cannot keep burning. It dies out due to lack of oxygen.
Carbon Dioxide Build-up
As the candle burns, it creates carbon dioxide gas. This gas fills the jar slowly. Carbon dioxide does not support burning. It replaces oxygen in the air around the flame. High carbon dioxide levels make it harder for the flame to get oxygen. The flame suffocates and goes out. The build-up of carbon dioxide is a silent killer for the flame.
Temperature And Pressure Changes
Putting a jar over a candle causes changes in temperature and air pressure inside the jar. These changes affect the candle’s flame and how long it burns. Understanding these effects helps explain why the flame behaves differently in this setup.
Heat Trapped Inside The Jar
The candle flame produces heat that stays inside the jar. This trapped heat raises the air temperature around the flame. Warm air expands and becomes less dense. It moves upward, creating a flow of hot gases inside the jar. The higher temperature can make the flame flicker or shrink.
Air Pressure Dynamics
As the candle burns, it uses oxygen from the air inside the jar. The oxygen level drops, and carbon dioxide increases. Burning also heats the air, causing it to expand. When the flame goes out, the air cools quickly. Cooler air contracts, lowering the pressure inside the jar. This pressure difference can pull liquid up or cause the jar to stick tightly to the surface below.
Common Misconceptions
Many people have ideas about what happens when you cover a candle with a jar. Some of these ideas are not quite right. It is important to understand the real science behind this simple experiment. This helps clear up confusion and shows what really causes the candle to go out.
Flame Sucking Air
Some think the flame pulls air inside the jar. They believe the candle uses up all the oxygen quickly. Then, the flame dies because there is no air left. This is only partly true. The flame does need oxygen to burn. But it does not suck air in like a vacuum. The candle burns the oxygen already inside the jar. Once the oxygen is low, the flame goes out.
Jar Creating A Vacuum
Others say the jar makes a vacuum that stops the flame. They think the cooling air inside the jar shrinks and pulls the flame down. This is not accurate. The jar does not create a strong vacuum. The flame dies mainly because of lack of oxygen. The air pressure changes inside the jar are very small. They do not affect the flame directly.
:max_bytes(150000):strip_icc()/lit-candles-in-mason-jars-on-table-681966283-5c47b57a46e0fb0001b89498.jpg)
Credit: www.thesprucecrafts.com
Practical Experiments To Try
Trying practical experiments with a candle and a jar helps you see science in action. These simple tests show how air and gases affect fire. You can learn about oxygen, pressure, and burning by doing these at home. Each experiment gives new insights. All you need are basic items like jars, candles, and sometimes water or other gases.
Different Jar Sizes
Use jars of various sizes to cover the candle. Notice how the flame changes with each jar. Smaller jars make the flame go out faster. Larger jars allow the candle to burn longer. This happens because bigger jars hold more oxygen. The candle uses oxygen to keep burning. Watching the flame die shows how important oxygen is.
Using Water Or Other Gases
Try filling the jar with water before placing it over the candle. The water blocks air from reaching the flame. The candle goes out quickly. You can also use other gases like carbon dioxide if safe. Different gases can stop the fire faster or slower. This experiment teaches about what fire needs to burn. It also shows how gases affect fire behavior.
Real-world Applications
Putting a jar over a candle is a simple act with clear, real-world uses. This experiment helps explain how fire needs air to burn. It also shows what happens to oxygen inside a closed space.
Many people use this idea to teach and learn about fire and air. It helps show dangers and safety tips in a clear way. The concept applies beyond just science lessons. It plays a role in daily safety and education.
Fire Safety Insights
Covering a candle with a jar stops the flame after a short time. The flame uses up the oxygen inside the jar. Without oxygen, the flame cannot stay alive. This simple fact teaches why fire needs air to burn.
Knowing this helps in fire safety. It explains why closing doors can stop a fire from spreading. It also shows why fresh air is needed to keep a fire going. People learn to avoid keeping fires in sealed spaces.
Scientific Demonstrations
This experiment is popular in classrooms and science shows. It visually proves that fire consumes oxygen. It also shows how gases behave in a closed container. Watching the flame die helps students understand air and fire.
Teachers use this to explain basic chemistry and physics. It makes learning active and fun. The clear, simple setup helps learners see science in action. This demonstration sparks curiosity about how fire and air work together.

Credit: rjallain.medium.com
Credit: www.acs.org
Frequently Asked Questions
What Happens To Candle Flame Under A Jar?
When a jar covers a candle, oxygen supply reduces. The flame uses up available oxygen and dims. Eventually, it goes out due to lack of oxygen needed for combustion.
Why Does Candle Flame Go Out In A Jar?
The candle flame extinguishes because the jar traps carbon dioxide. Oxygen inside depletes as the candle burns. Without oxygen, the flame cannot sustain combustion and dies out.
How Does Air Affect A Candle In A Jar?
Air contains oxygen essential for burning. When a jar covers a candle, oxygen inside decreases. Less oxygen causes the flame to weaken and eventually extinguish.
Can A Candle Burn Without Oxygen Inside A Jar?
No, a candle cannot burn without oxygen. The jar limits oxygen supply. Once oxygen is used up, the flame stops burning.
Conclusion
Placing a jar over a candle changes how the flame behaves. The flame uses oxygen inside the jar and then goes out. This shows how fire needs air to keep burning. It also helps us understand basic science about air and fire.
Next time you see a candle, try this safe experiment. It’s a simple way to learn about air and flames. Always be careful and never leave a burning candle alone. This small test teaches a big lesson about fire and air.
